ACCESS:
Information on how to use these systems is available at the free training seminar given once per month. Attendance is mandatory prior to beginning one-on-one training and there are no exceptions so please plan accordingly. Sign up for the training seminar using the "Contact Us" link at the top of the page and include the phrase "Confocal Training Seminar", in the comments box. Your registration is complete and you will not receive a confirmation email. This seminar is followed by hands-on, individual instruction.
RATES:
Please refer to our rates page.
INFORMATION:
We have three confocal systems available for independent use:
OLYMPUS FV 3000 CONFOCAL SYSTEM: This inverted scanning confocal offers high resolution imaging and high sensitivity fluorophore detection in fixed cells and tissues either on slides or in well plates or petri dishes. Much faster but lower resolution imaging using a resonant scanner is also an option. The system is equipped with 3 air and 3 oil objectives up to 60x oil and 5 fluorescence detectors, two of which are extra high sensitivity. The system uses 5 solid diode lasers ranging from 405 to 752 nm. For detailed technical information regarding this system, refer to this PDF.

OLYMPUS FV1000 FILTER CONFOCAL SYSTEM: offers routine confocal microscopy of fixed tissue sections and cultured cells. It is equipped with four lasers, four fluorescence detectors and several high N.A. and short working distance objectives. For detailed technical information regarding this system refer to this PDF.
Confocal laser scanning microscopy permits high resolution optical sectioning without compromising specimen integrity. Some advantages of using confocal microscopy include:
- Elimination of out of focus light, yielding sharper images than conventional fluorescence (widefield) microscopy.
- Analysis of thick specimens.
- Computer controlled microscopy, permitting fine focus control and digitization of images.
- Collection of data sets for subsequent three dimensional reconstruction.
- Ability to collect several images at the same time from specimens labelled with multiple fluorochromes.
- Improved detection of protein colocalization over conventional widefield microscopy.
SAMPLE IMAGES:
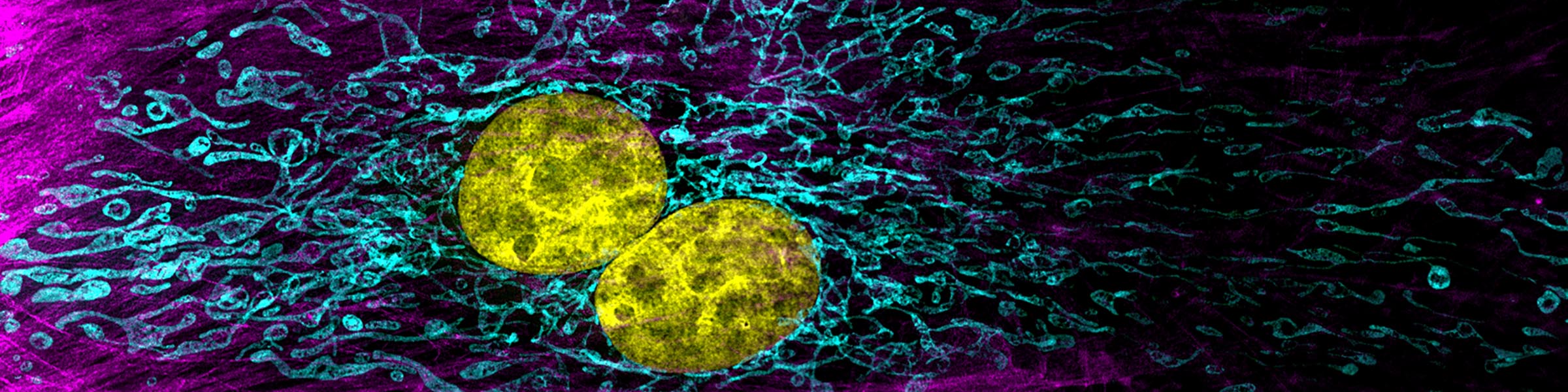
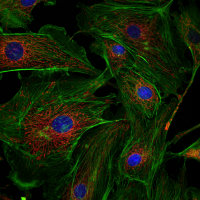 Cells in culture - Z projection of image stack
Cells in culture - Z projection of image stack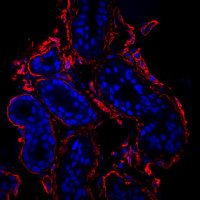 Cells in culture - Skin section Z projection of image stack
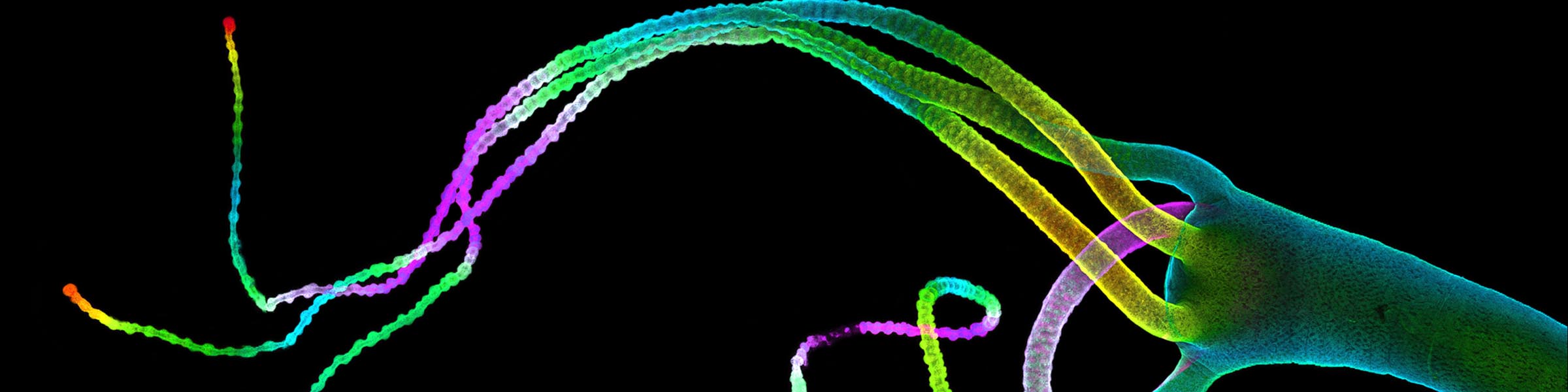
Cells in culture - Skin section Z projection of image stack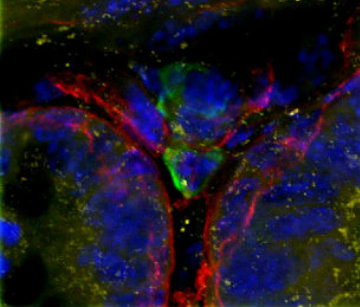 Cells in culture - 4 color imaging
Cells in culture - 4 color imaging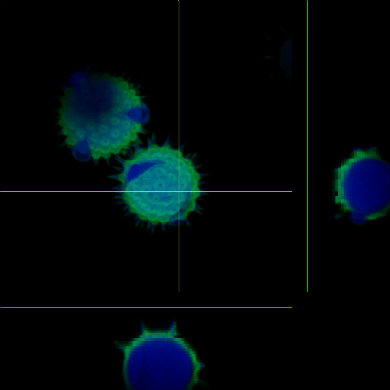 Orthagonal view of pollen
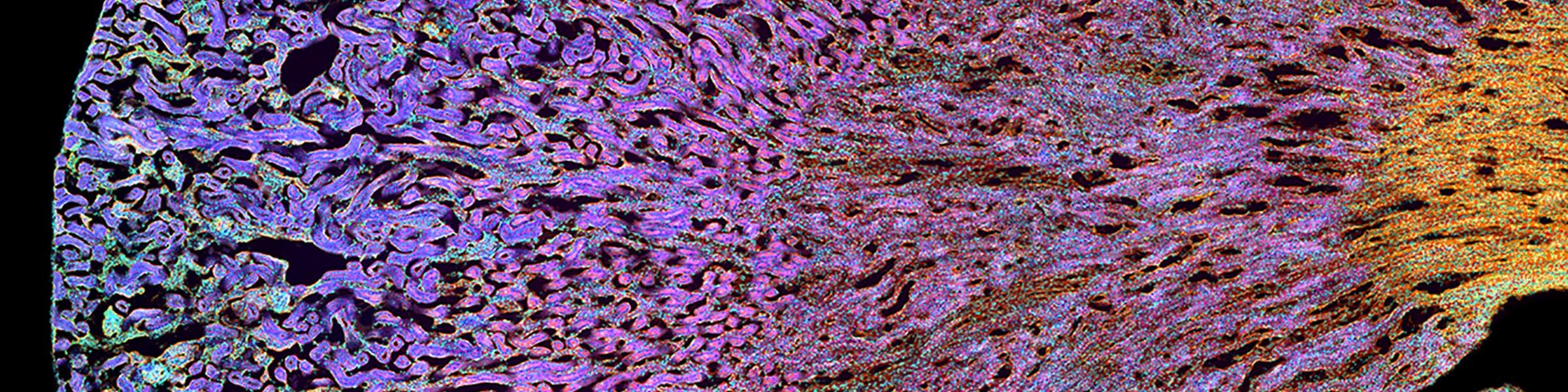
Orthagonal view of pollen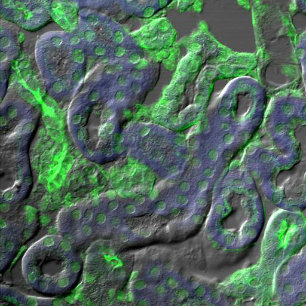 Fluorescence overlay with DIC
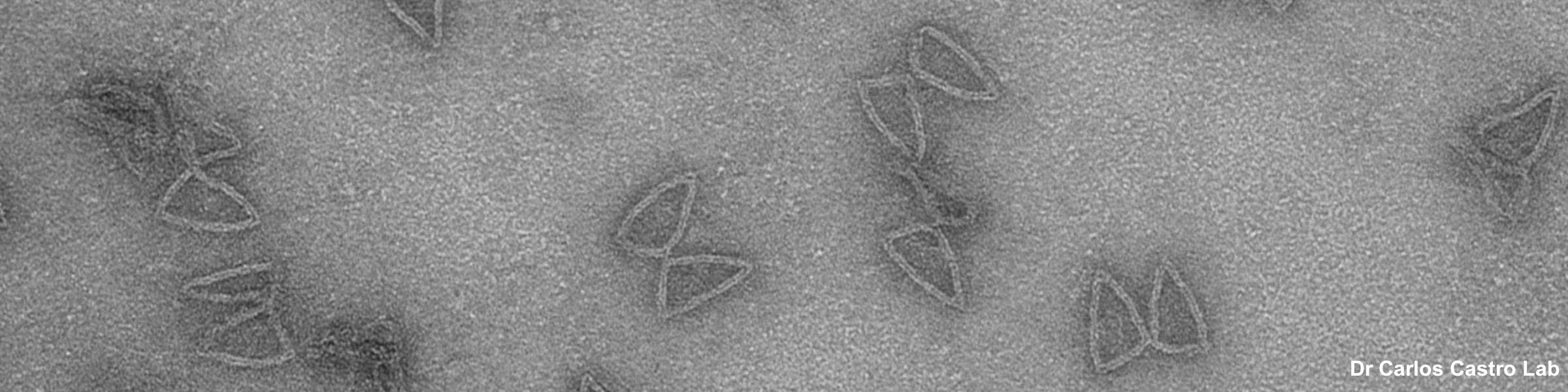
Fluorescence overlay with DIC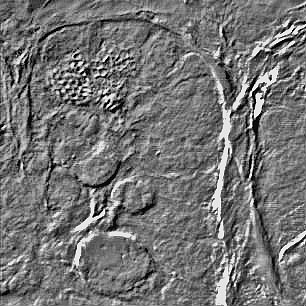 DIC image of tissue sample
DIC image of tissue sample